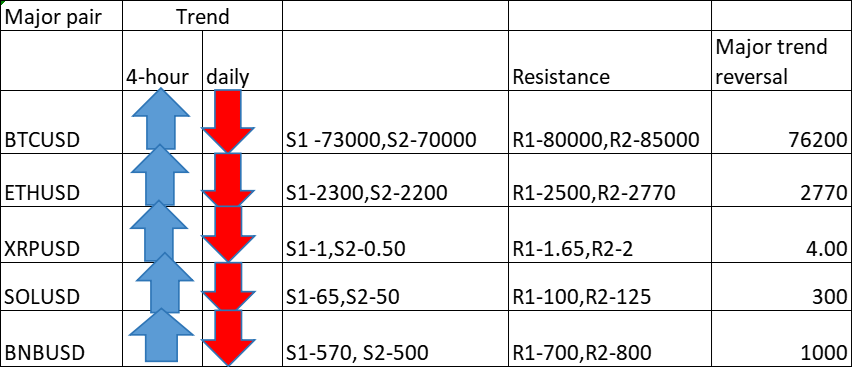
Mar 21, 2017 11:02 am UTC| Business
- EMA guidance provides clear regulatory pathway for EU approval for Iomab-B - Trial design, primary endpoint and planned statistical analysis from the Iomab-B U.S. pivotal Phase 3 SIERRA trial are acceptable as the...
OncoCyte Submits Application for CLIA Certification of its Laboratory
Mar 21, 2017 11:02 am UTC| Business
ALAMEDA, Calif., March 21, 2017 -- OncoCyte Corporation (NYSE MKT:OCX), a developer of novel, non-invasive blood based tests to aid in the early detection of cancer, has submitted its application for CLIA certification...
OncoCyte Submits Application for CLIA Certification of its Laboratory
Mar 21, 2017 11:02 am UTC| Business
ALAMEDA, Calif., March 21, 2017 -- OncoCyte Corporation (NYSE MKT:OCX), a developer of novel, non-invasive blood based tests to aid in the early detection of cancer, has submitted its application for CLIA certification...
OncoCyte Submits Application for CLIA Certification of its Laboratory
Mar 21, 2017 11:02 am UTC| Business
ALAMEDA, Calif., March 21, 2017 -- OncoCyte Corporation (NYSE MKT:OCX), a developer of novel, non-invasive blood based tests to aid in the early detection of cancer, has submitted its application for CLIA certification...
Mar 21, 2017 11:01 am UTC| Business
ELISPOT results suggest that HS-110 plays an integral role in tumor reduction and may enhance efficacy of checkpoint inhibitors in lung cancer patients Tumor reduction corresponds to enhanced immune response DURHAM,...
Mar 21, 2017 11:01 am UTC| Business
ELISPOT results suggest that HS-110 plays an integral role in tumor reduction and may enhance efficacy of checkpoint inhibitors in lung cancer patients Tumor reduction corresponds to enhanced immune response DURHAM,...
Mar 21, 2017 11:01 am UTC| Business
TUCSON, Ariz., March 21, 2017 -- HTG Molecular Diagnostics, Inc. (Nasdaq:HTGM) (“HTG”), a provider of instruments, reagents, and services for molecular profiling applications, today announced that the Company will...
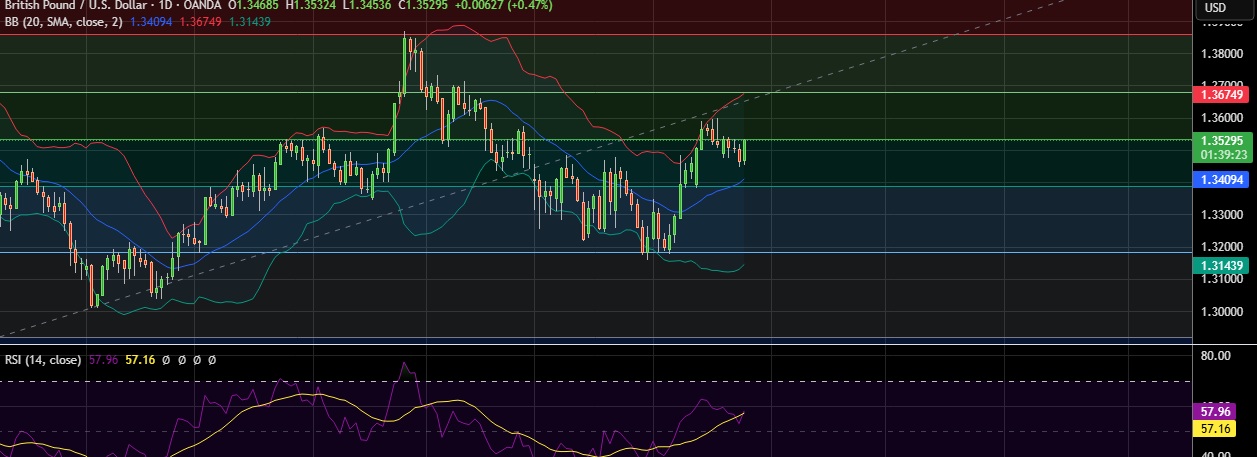
- Market Data