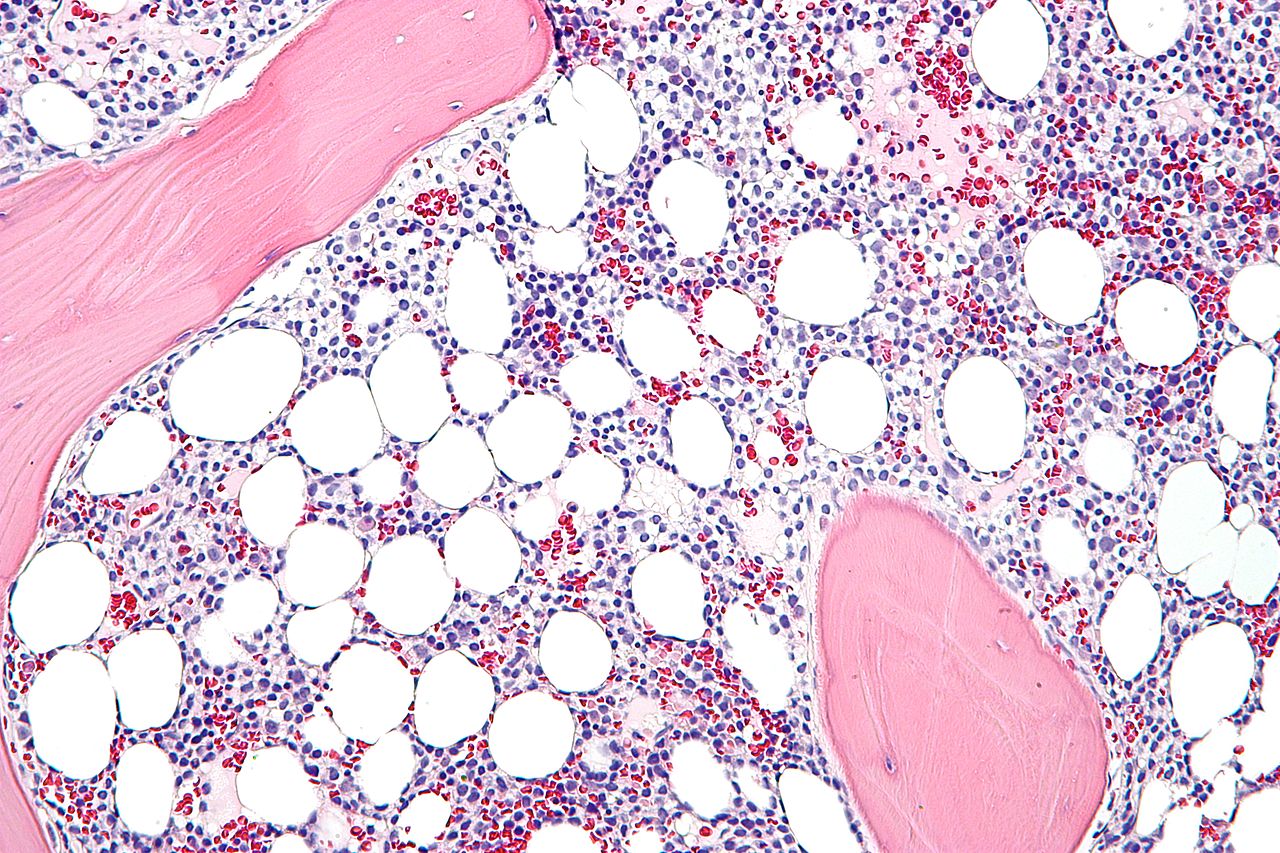
It’s a new age for science as the US Food and Drug Administration gave its approval for scientists to conduct genetic manipulation in an effort to combat leukemia. This could potentially lead to a cure that does not require extensive chemotherapy, but it is also a new frontier. Gene-editing technology has existed for decades but it wasn’t until recently that it became much cheaper and more accessible.
For all intents and purposes, genetic manipulation can be used for practically any end for as long as it has something to do with organic materials. In the case of humans, the tool called CRISPR can allow scientists to finally start cutting away or adding specific genetic materials that can change a person’s physiology. With regards to leukemia, scientists want to alter cells to make them more resistant to the disease, the New York Times reports.
By creating tougher and more aggressive cells to fight leukemia or other forms of cancer, patients would theoretically never have to take medicine of any kind ever again. They would have a living drug in their system, which would make them practically immune to fatal diseases.
Naturally, there are plenty of concerns when it comes to genetic manipulation that makes getting approval for tests incredibly difficult. The company that finally got approval is Novartis and one of the company’s biggest achievements is 12-year-old Emily Whitehead who is now free of leukemia thanks to the revolutionary treatment.
When speaking to the FDA panel before approval was granted, Emily’s father, Tom Whitehead made a passionate case in favor of the treatment. The premise was simple; approve the treatment and thousands will be saved, Futurism reports.
“I hope that someday all of you on the advisory committee can tell your families for generations that you were part of the process that ended the use of toxic treatments like chemotherapy and radiation as standard treatment, and turned blood cancers into a treatable disease that even after relapse most people survive.”



 Vanda Pharmaceuticals (VNDA) Stock Soars After FDA Approves BYSANTI for Bipolar I and Schizophrenia
Vanda Pharmaceuticals (VNDA) Stock Soars After FDA Approves BYSANTI for Bipolar I and Schizophrenia  Novo Nordisk Shares Tumble After CagriSema Misses Key Trial Endpoint
Novo Nordisk Shares Tumble After CagriSema Misses Key Trial Endpoint  Daiichi Sankyo Stock Drops After Earnings Delay and Oncology Review
Daiichi Sankyo Stock Drops After Earnings Delay and Oncology Review  Organon Stock Surges After Reports of $13 Billion Buyout Bid by Sun Pharma
Organon Stock Surges After Reports of $13 Billion Buyout Bid by Sun Pharma  U.S. Officials Express Optimism Over New CDC Director Selection Amid Vaccine Policy Turmoil
U.S. Officials Express Optimism Over New CDC Director Selection Amid Vaccine Policy Turmoil  RFK Jr. Faces Scrutiny Over David Geier’s HHS Role and Vaccine Review Work
RFK Jr. Faces Scrutiny Over David Geier’s HHS Role and Vaccine Review Work  Novartis’ Vanrafia Shows Strong Phase 3 Results in IgA Nephropathy, Paving Way for Full Approval
Novartis’ Vanrafia Shows Strong Phase 3 Results in IgA Nephropathy, Paving Way for Full Approval  The four types of dementia most people don’t know exist
The four types of dementia most people don’t know exist  CDC Acting Director Urges Measles Vaccination as U.S. Cases Surge in 2026
CDC Acting Director Urges Measles Vaccination as U.S. Cases Surge in 2026  Novo Nordisk Raises 2026 Outlook on Strong Wegovy Demand
Novo Nordisk Raises 2026 Outlook on Strong Wegovy Demand  Innovent Biologics Shares Rally on New Eli Lilly Oncology and Immunology Deal
Innovent Biologics Shares Rally on New Eli Lilly Oncology and Immunology Deal  Moderna Stock Drops After FDA Declines Review of mRNA Flu Vaccine
Moderna Stock Drops After FDA Declines Review of mRNA Flu Vaccine  Moderna to Pay Up to $2.25B to Settle LNP Patent Dispute Over COVID-19 Vaccine Technology
Moderna to Pay Up to $2.25B to Settle LNP Patent Dispute Over COVID-19 Vaccine Technology