SAN DIEGO, July 19, 2016 -- Access Scientific has introduced the POWERWAND™ XL, a companion to the POWERWAND ALL-IN-ONE. The new POWERWAND XL is designed specifically for clinicians who prefer to use a component-based, over-wire insertion technique similar to the Modified Seldinger Technique (MST) they currently use. The XL technique feels similar to the MST but has these distinct advantages:
- Improves clinical outcomes through atraumatic catheter insertion
- Designed to reduce risk of deep-vein thrombosis by up to 500%; no trimming of catheter necessary
- Reduces risk of back leaking; no peel-away sheath
- Guidewire extends fully so the catheter does not drag across the intima of the vessel
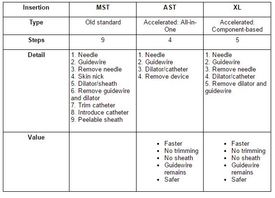
- Faster and safer because of fewer steps (see table below).
|
||||||||||
As with MST insertions, the XL insertion technique utilizes a 21-gauge safety needle and guidewire to access the vessel. The XL accelerates line placement and makes it safer compared to MST, by eliminating the need for a peelable introducer sheath. Instead, the proprietary POWERWAND XL dilator and catheter are inserted directly over the wire and into the bloodstream. This technique eliminates the sheath-related steps in MST that can be associated with such risks as bleeding, back-leaking, and vessel damage.
The ALL-IN-ONE is inserted with the Accelerated Seldinger Technique (AST), which also improves on the MST in speed, number of steps, and safety (see table below). Both ALL-IN-ONE and XL deliver the evidence based, best in class midline:
- Size: 4Fr and 5Fr
- Length: 8 and 10cm
- Infection Prevention: ZERO BSIs over 12,000 catheter-days
- High Flow: Capable of delivering large volumes rapidly, 130-180 ml/min
- Power Injectable: 325psi / 8ml per second
- Blood Drawable: Allows for blood draws in most patients, 60 – 99%
- Patient Satisfaction: Provides patients with one stick hospitalization
Insertion Technique Comparison: MST, AST, and XL
[table #1]
About Access Scientific, LLC
Access Scientific is dedicated to the development and commercialization of breakthrough, proprietary devices that make vascular access safer for patients and reduce the cost of care. The company pioneered the power-injectable midline market with the POWERWAND™. Made of proprietary ChronoFlex™ C, the POWERWAND midline has been the proven, evidence based, best-in-class, with over 12,000 catheter-days of published scientific data attesting to the lowest complication rate and highest completion-of-therapy rate of any vascular access device ever studied. The POWERWAND offers a unique approach to reducing hospital-acquired infections.
Photos accompanying this release are available at:
http://www.globenewswire.com/newsroom/prs/?pkgid=40876
http://www.globenewswire.com/newsroom/prs/?pkgid=40877
CONTACT: Liz Dowling, (415) 388-2794
Dowling & Dennis Public Relations
E-mail: [email protected]



 Lumentum Holdings Rides AI Wave With Order Book Filled Through 2028
Lumentum Holdings Rides AI Wave With Order Book Filled Through 2028  Kia Cuts EV Sales Target for 2030 Amid Slowing Demand and U.S. Policy Shifts
Kia Cuts EV Sales Target for 2030 Amid Slowing Demand and U.S. Policy Shifts  Goldman Sachs, ANZ Cut Oil Forecasts Amid U.S.-Iran Ceasefire Hopes
Goldman Sachs, ANZ Cut Oil Forecasts Amid U.S.-Iran Ceasefire Hopes  Chalco Stock Surges as Q1 2025 Profit Forecast Jumps Up to 58%
Chalco Stock Surges as Q1 2025 Profit Forecast Jumps Up to 58%  Disney Plans to Cut 1,000 Jobs Amid Ongoing Restructuring Efforts
Disney Plans to Cut 1,000 Jobs Amid Ongoing Restructuring Efforts  Pilots Fear Retaliation for Refusing Middle East Flights Amid Ongoing Conflict
Pilots Fear Retaliation for Refusing Middle East Flights Amid Ongoing Conflict  China Vanke Seeks Bond Extension Amid Mounting Debt Crisis
China Vanke Seeks Bond Extension Amid Mounting Debt Crisis  China's AI Stocks Surge as Zhipu and MiniMax Hit Record Highs
China's AI Stocks Surge as Zhipu and MiniMax Hit Record Highs  Pony.ai, Uber, and Verne Launch Europe's First Commercial Robotaxi Service in Zagreb
Pony.ai, Uber, and Verne Launch Europe's First Commercial Robotaxi Service in Zagreb  Bendigo and Adelaide Bank Posts Strong Q3 Earnings, Announces AI-Driven Job Cuts
Bendigo and Adelaide Bank Posts Strong Q3 Earnings, Announces AI-Driven Job Cuts  NIO ES9 SUV Launch Sends HK Shares Down 7% Despite Bold Pricing Strategy
NIO ES9 SUV Launch Sends HK Shares Down 7% Despite Bold Pricing Strategy  TSMC Posts Strong Q1 2025 Revenue, Riding AI Chip Demand Wave
TSMC Posts Strong Q1 2025 Revenue, Riding AI Chip Demand Wave  U.S. Automakers Push Back Against EU Rules Blocking American Trucks from European Market
U.S. Automakers Push Back Against EU Rules Blocking American Trucks from European Market  San Francisco Suspect Arrested After Molotov Cocktail Attack on OpenAI CEO Sam Altman's Home
San Francisco Suspect Arrested After Molotov Cocktail Attack on OpenAI CEO Sam Altman's Home  SanDisk Joins Nasdaq-100, Replacing Atlassian on April 20
SanDisk Joins Nasdaq-100, Replacing Atlassian on April 20  Anthropic's Mythos AI Model Sparks Emergency Cybersecurity Meeting With Top U.S. Bank CEOs
Anthropic's Mythos AI Model Sparks Emergency Cybersecurity Meeting With Top U.S. Bank CEOs  Abbott Laboratories Ordered to Pay $53 Million in Premature Infant Formula Lawsuit
Abbott Laboratories Ordered to Pay $53 Million in Premature Infant Formula Lawsuit